Information for this Appendix is provided by Dr R B Chavan
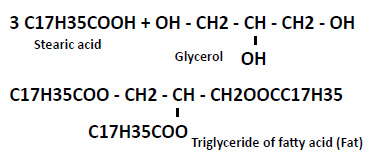
Fats
Fats are triglycerides of fatty acids. The formation of fats is illustrated from the reaction between 3 molecules stearic acid with 1 molecule of glycerol.
Waxes
Waxes are esters of fatty acids formed by the reaction between fatty acid and fatty alcohol. For example the reaction between stearic acid and montanyl alcohol to produce wax

The natural fatty substances in cotton ball serve as a protective barrier both to water penetration and to microbial degradation of cotton fibre during its growth on cotton plant. Fatty substances also act as lubricant that is essential for proper spinning of cotton fibre into yarn. However, fatty substances are not desirable in the chemical processing and finishing of the cotton yarns and fabrics because they interfere with wetting of the fibre and penetration of chemicals and dyes. Thus incomplete removal of fatty substances during pre-treatment processes is responsible for poor absorbency of cotton fabric resulting in uneven dyeing or printing.
Pectins
Pectin’s present in cotton are derivatives of pectic acid. Pectic acid is the polymer of high mol wt composed of galactouronic acid whose chemical structure is shown below:
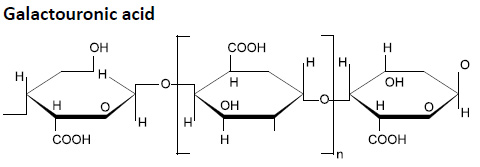
Some of the COOH groups present in galactouronic acid are present in the form of Ca and Mg salts. Pectic acid with free COOH and their Ca and Mg salts is insoluble in water.
Proteins
Proteins present in cotton are nitrogenous compounds with amide (-CONH) groups. Proteins are formed by the reaction between amino acids and fatty acids.
Colouring matter
The cream colour of cotton is due to presence of two coloured pigments. The chemical structures of these
pigments are as follows:
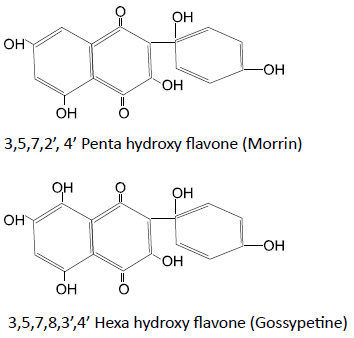
Presence of pectin’s, proteins and colouring matter are responsible of cream colour of grey cotton fabric. It is necessary to remove these impurities to improve the whiteness of cotton fabric and brightness of colours after dyeing and printing.
Leave a Reply